OUR
SERVICES
FROM DESIGN TO PUBLICATIONS
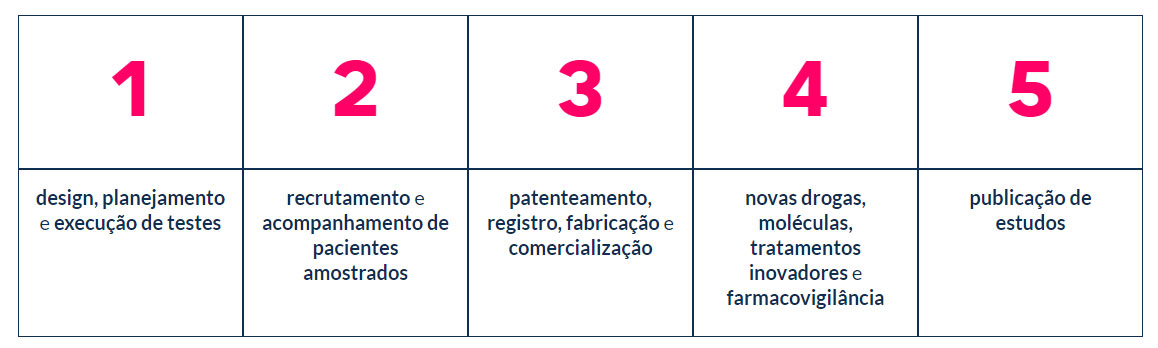
The services range from study design to the development and approval of study protocols, through planning and check points with sponsor, pre-clinical and clinical research (phases 1 to 4), biopharmaceutical development, development of biological assays, monitoring preventive, clinical trial management, data/study management, pharmacovigilance, technical-regulatory support in various approvals, drug dispensing and publications of articles and studies. Science Valley works in an integrated manner in the clinical trial processes that will serve as a basis for the following steps:
REMARKABLE EXPERIENCE
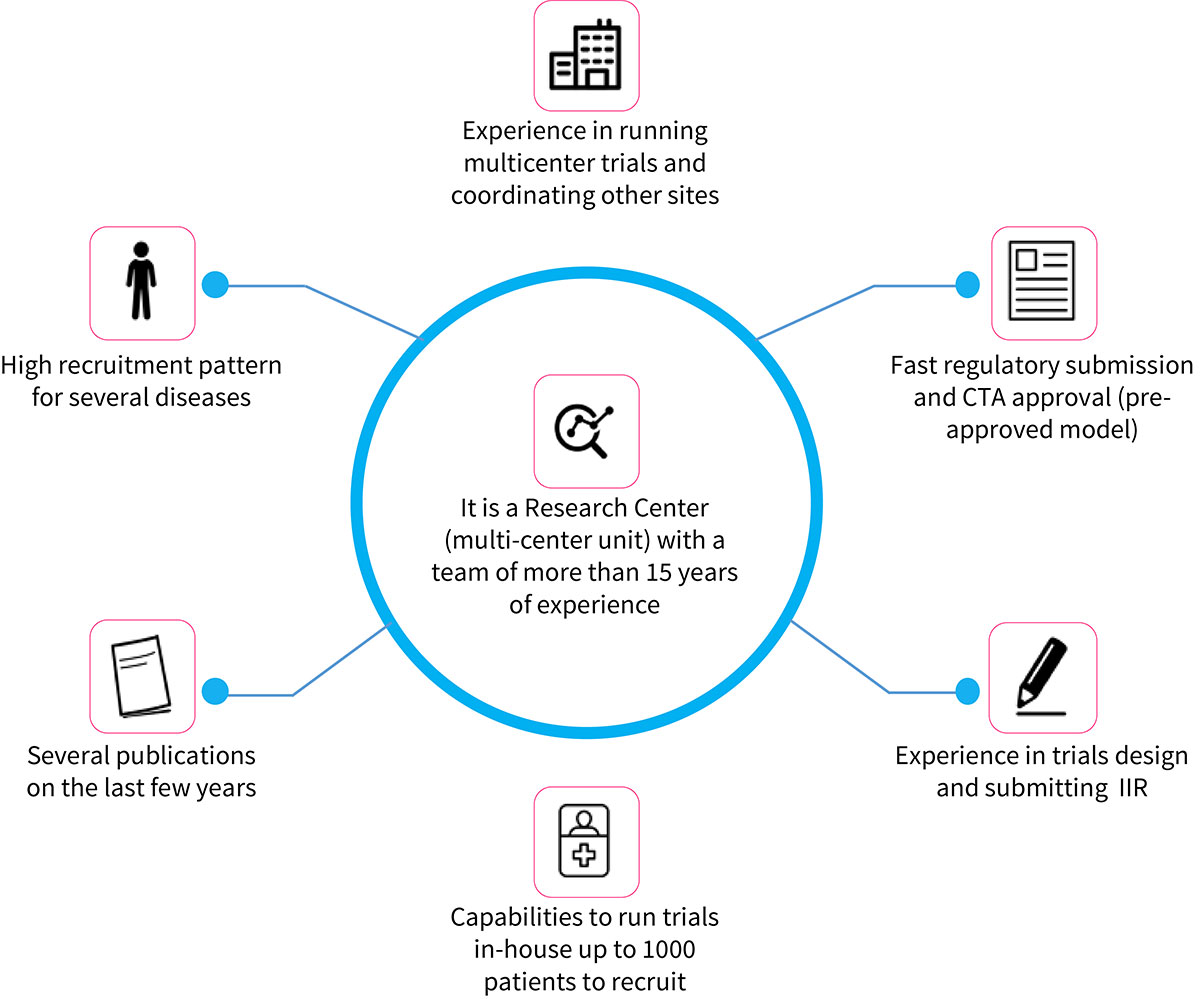
Science Valley has experience in the field that gives you confidence in carrying out your research:
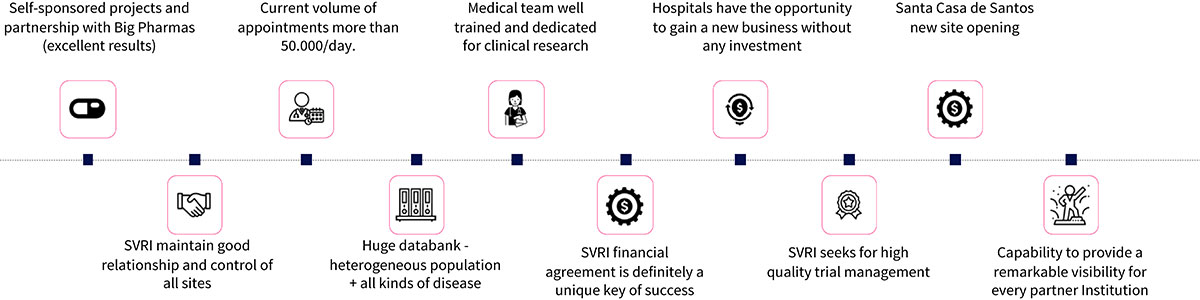
HOW WE WORK
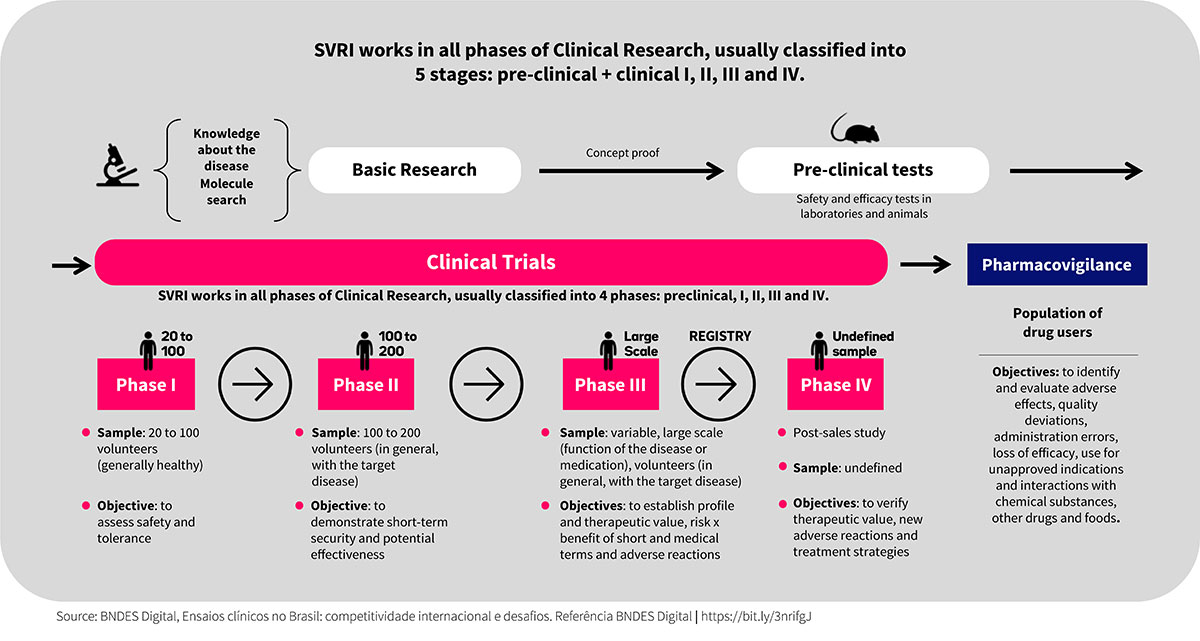
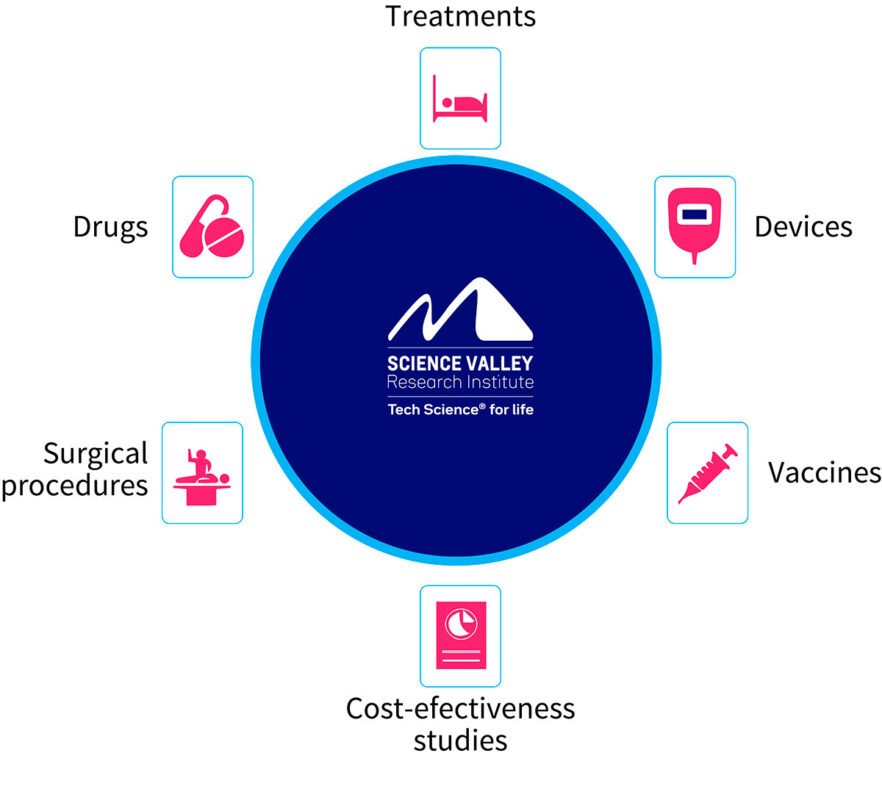
WHAT WE DELIVER
With international technical quality standards required and permanently auditable, Science Valley operates with a multidisciplinary team and high medical-scientific capacity, operational technology that follows standards in line with Good Clinical Practices (GCP) and the Brazilian General Personal Data Protection Law. Science Valley is able to test, through its integrated clinical research.
OUR DIFFERENTIALS