OUR
GOVERNANCE
CORPORATE GOVERNANCE
The Science Valley’s governance matrix supports the company’s strategic objectives, operating with a trained team, made up of health science specialists and professionals. Governance ensures technological processes aligned with the most demanding international standards of “Good Clinical Practice (GCP)”.
SECURITY AND RELIABILITY
Conducting all R&D processes with security in data processing, ensuring high quality in the recruitment and monitoring of the patient panel and trust for direct or indirect stakeholders in the contracted services, considering of all guidelines related to regulatory agents, such as Food and Drug Administration (FDA) and Agência Nacional de Vigilância Sanitária (ANVISA).
ETHICS AND COMPLIANCE
Commitment to look after the health and quality of life of patients, always committed to following the strictest ethical standards and transparency, based on ethical, permanent and irrevocable foundations. Science Valley works with a Compliance platform that supports the company’s strategic objectives through compliance with the law, public policy and the health regulatory system. This platform is composed of:
- Law and governance
- GCP and research practice
- Patient protection
- No conflict of interest
- Informed Consent to research
- Respect for confidentiality
QUALITY ASSURANCE
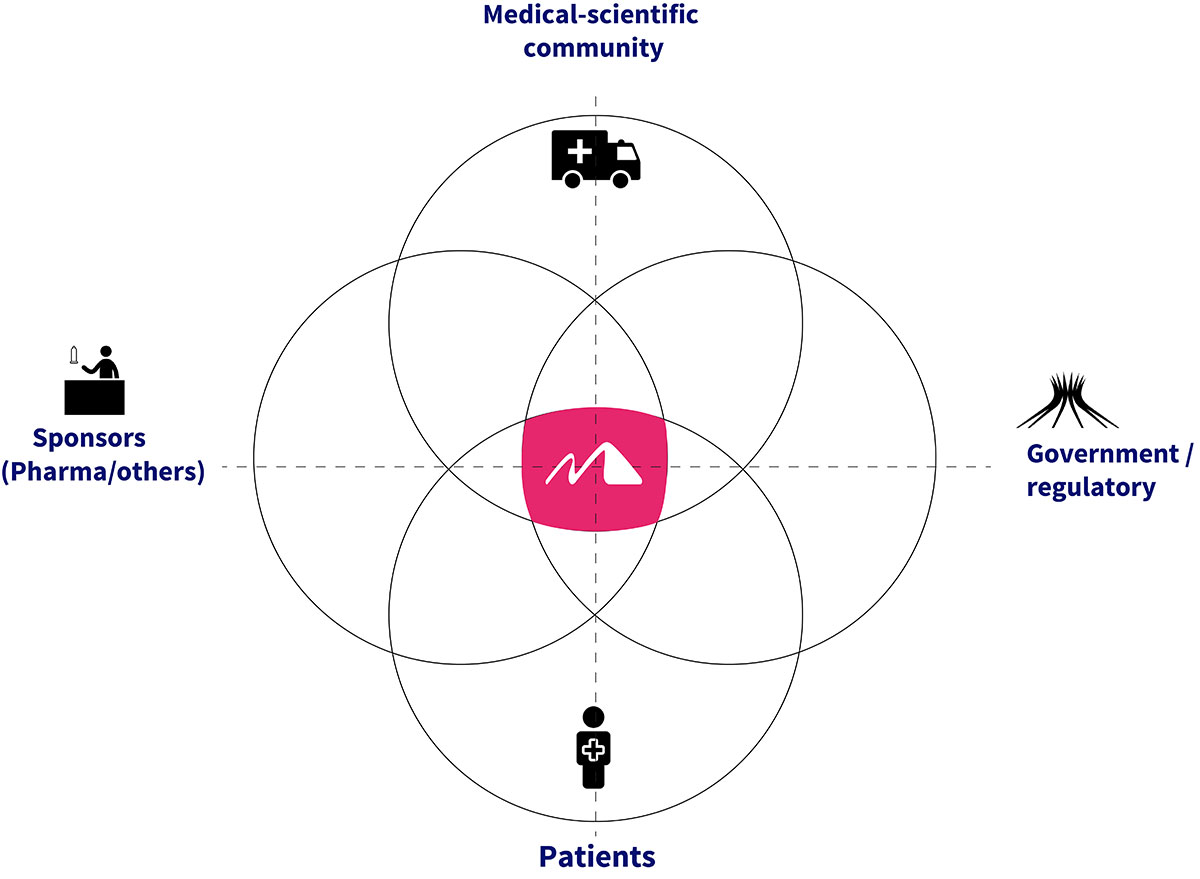
Unprecedent multicentric management of clinical research as a historical competitive differential. The company’s business model allows it to meet the standards of ethics and governance in its R&D processes linked to four areas of interest (medical-scientific community/hospitals, sponsors, regulatory agents and patients), which work in an integrated manner for the study design, execution and delivery of requested clinical trial results: